The release date for generic Cialis has been confirmed for late 2023. Patients seeking an affordable alternative to the brand-name medication will soon have access to options that provide similar effectiveness in treating erectile dysfunction. This new development promises to improve accessibility for many who rely on these treatments.
Manufacturers are preparing to launch their generic versions, ensuring they meet the stringent quality and safety standards expected in the pharmaceutical industry. With multiple companies entering the market, increased competition may lead to lower prices, benefiting consumers looking for budget-friendly solutions.
As the date approaches, it’s advisable to consult with your healthcare provider about transitioning to a generic formulation. Staying informed about dosage and potential side effects ensures a smooth shift to these new options without compromising health and well-being.
- Release Date for Generic Cialis
- Availability and Pricing
- Consultation and Prescription
- Overview of Cialis and Its Generic Version
- Dosage and Administration
- Side Effects and Precautions
- Current Market Status of Generic Cialis
- Expected Release Timeline for Generic Cialis
- Current Status and Approval Process
- Market Availability Projections
- Potential Impact on Prices and Availability
- Regulatory Approvals and Considerations
- Post-Approval Monitoring
- International Approvals
Release Date for Generic Cialis
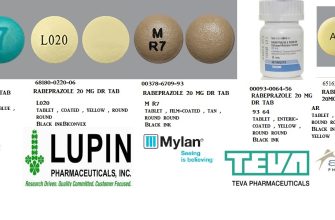
The generic version of Cialis, known as tadalafil, was approved by the FDA and became available in the United States on December 11, 2017. Patients seeking a more affordable option found this release beneficial, as it significantly reduced the price compared to the brand-name product.
Availability and Pricing
Many pharmacies now stock generic tadalafil, making it easily accessible. The competition among manufacturers has contributed to lower prices on the market. Consumers can expect to pay considerably less than the original Cialis, enhancing affordability without sacrificing quality.
Consultation and Prescription
Before starting tadalafil, discuss it with a healthcare provider to determine if it’s suitable for your condition. A prescription is necessary for obtaining this medication, ensuring safe and appropriate usage. Your doctor can provide guidance regarding dosage and any potential interactions with other medications.
Overview of Cialis and Its Generic Version
Cialis, containing the active ingredient tadalafil, treats erectile dysfunction and symptoms of benign prostatic hyperplasia. It enhances blood flow to the penis, facilitating an erection during sexual stimulation. The effects can last up to 36 hours, offering flexibility in timing for intimacy.
The generic version of Cialis is tadalafil, which is available at a lower cost. It is chemically identical to the brand-name medication and provides the same therapeutic benefits. Patients often find the generic option appealing due to affordability without compromising effectiveness. Both versions require a prescription and should be used under medical guidance.
Dosage and Administration
For erectile dysfunction, Cialis can be taken as needed or in a lower daily dose. The on-demand dose typically ranges from 10 mg to 20 mg, taken at least 30 minutes before sexual activity. Daily use involves a standard dose of 2.5 mg to 5 mg, enabling spontaneous sexual activity without planning. Adhering to prescribed dosages ensures optimal results and minimizes side effects.
Side Effects and Precautions
Common side effects may include headaches, indigestion, back pain, and flushing. Serious side effects require immediate medical attention. Men with certain health conditions, such as severe heart problems or those taking nitrates, should avoid tadalafil. Consulting a healthcare provider before starting treatment helps identify potential interactions with other medications and ensures safety.
Current Market Status of Generic Cialis
Generic Cialis, known for its active ingredient tadalafil, has gained significant traction in the pharmaceutical market. The generic version offers an affordable alternative to the brand-name product, benefiting many consumers looking for effective treatment for erectile dysfunction.
The market saw the introduction of generic Cialis following the expiration of the original product’s patent. This opened doors for several manufacturers to produce and distribute tadalafil, resulting in increased availability and competitive pricing.
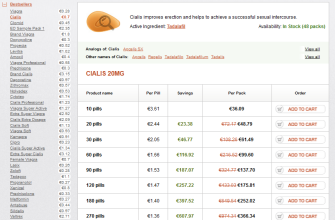
Pricing for generic Cialis varies, largely depending on the manufacturer and the pharmacy. On average, consumers can expect to pay significantly less than for the brand-name version. Many pharmacies offer discounts and coupons, further reducing out-of-pocket costs.
Availability remains high across various regions. Both brick-and-mortar pharmacies and online platforms stock generic Cialis, making it accessible to a broader audience. Legitimacy is crucial; purchasers should always verify that the online retailers comply with regulatory standards.
Current competition includes several established pharmaceutical companies that have entered the market with their branded generics. This competitive environment fosters innovation and leads to improved consumer offerings. Regular updates on manufacturer approvals help ensure that consumers have safe options to choose from.
Below is a table summarizing key points about the current market status of generic Cialis:
| Aspect | Status |
|---|---|
| Availability | High across pharmacies and online retailers |
| Average Price | Lower than brand-name, discounts available |
| Competition | Multiple manufacturers, fostering innovation |
| Regulatory Compliance | Varies; check legitimacy of online sources |
| Consumer Awareness | Growing awareness of generic options |
This market status reflects a robust landscape for generic Cialis, providing consumers with both choice and affordability in their healthcare options. Continuous monitoring of prices and available manufacturers will benefit those seeking the best value for their purchase.
Expected Release Timeline for Generic Cialis
The generic version of Cialis is anticipated to enter the market in the next few years, providing an affordable alternative for those seeking treatment for erectile dysfunction. The patent for the branded drug, which is manufactured by Eli Lilly, has already expired. However, the approval process for generic medications can take time.
Current Status and Approval Process
A few pharmaceutical companies have applied for the generic Cialis approval. The U.S. Food and Drug Administration (FDA) is reviewing these applications. While the timeline can vary, it generally spans several months to a few years from submission to approval. Key steps include:
- Submission of abbreviated new drug applications (ANDAs) to the FDA.
- Review of clinical data to ensure safety and efficacy.
- Final approval and market entry following successful review.
Market Availability Projections
With the pace of the approval process in mind, many experts predict that generic Cialis could become available by 2025. Market conditions and competition among manufacturers will further influence accessibility and pricing. Once released, patients will benefit from lower costs, making treatment more accessible.
To stay updated on the release, consider monitoring FDA announcements or consulting healthcare providers for the latest information on available generic options.
Potential Impact on Prices and Availability
The introduction of generic Cialis is likely to lead to a significant reduction in prices. Once the patent expires, multiple manufacturers can produce the drug, fostering competition. This heightened competition typically drives down prices, making it more accessible for consumers. Surveys indicate that generics can cost up to 80% less than their brand-name counterparts.
Availability will also improve. With several companies entering the market, pharmacies will stock a broader range of options. This can result in fewer stock shortages and better access for patients. Many online pharmacies are already preparing for the influx of generic versions, enhancing convenience for those seeking discreet purchases.
Healthcare providers may recommend generics, as they often provide the same active ingredients and effectiveness as the brand-name drug. As awareness increases, patients will likely see generics as a reliable alternative, further boosting their usage in the market.
Additionally, insurance providers may adjust their coverage policies to promote generics, potentially lowering out-of-pocket costs for consumers. This shift can be particularly beneficial for those with chronic conditions requiring ongoing treatment.
Monitoring local pricing and availability will be essential. Using comparison websites can help consumers find the best deals. Staying informed about the release of generics ensures that patients are well-equipped to make economical choices regarding their healthcare.
Regulatory Approvals and Considerations
Generic Cialis, containing the active ingredient tadalafil, must secure regulatory approval from agencies like the FDA before its market release. Manufacturers submit data on safety, efficacy, and quality through a New Drug Application (NDA) or Abbreviated New Drug Application (ANDA) for generics. This process includes rigorous clinical trials to demonstrate that the generic formulation is bioequivalent to the brand-name drug.
Once an ANDA is submitted, the review cycle can last several months. During this time, regulatory bodies assess the data and may request additional information, which can further delay timelines. Companies should anticipate these hurdles and prepare thorough documentation to streamline the approval process.
Post-Approval Monitoring
After approval, ongoing monitoring continues. Adverse events must be reported by manufacturers to ensure the long-term safety of the generic option. Surveillance programs take place to assess the medication’s performance in the broader population, ensuring that any potential issues are addressed swiftly.
International Approvals
For global distribution, companies must also seek approvals in different jurisdictions. Each country has its own regulatory body, such as the EMA in Europe or TGA in Australia. Sticking to regulatory guidelines in each locale is crucial, as variations in requirements can impact release dates and market strategies.